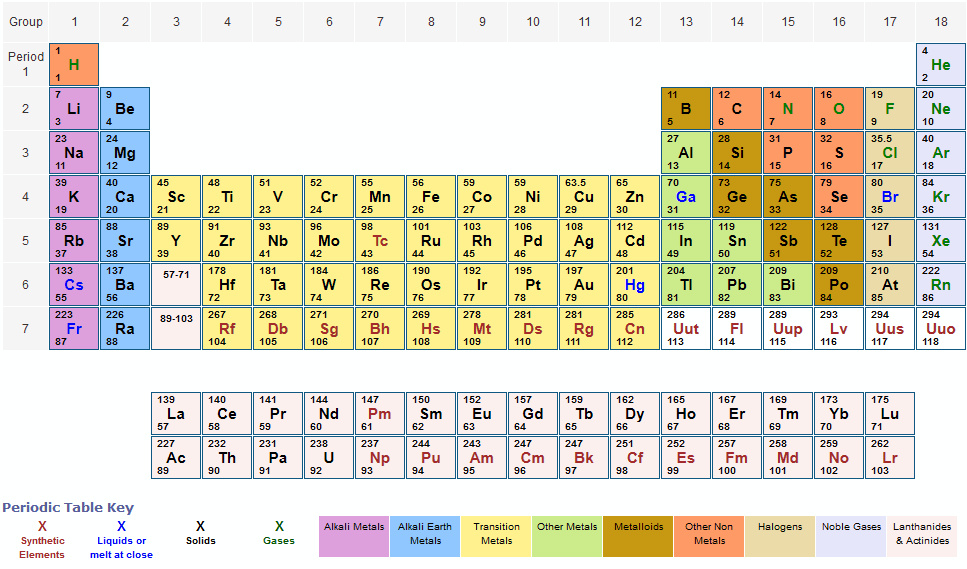
And don’t forget to put the unit g/mol to your final calculated molar mass.First solve the brackets, then multiplications and at last do the final addition. Options for hiding the symbol or name of the elements provide a handy learning aid for memorizing the periodic table. 
Always follow the calculation order to avoid any mistakes in calculation. Use this periodic table for calculating molar mass for any chemical formula.But all these units (i.e g/mol, grams/mole and g/mole) are the same. In some books, you may see the unit of molar mass as grams/mole or g/mole.Now here we have to find the molar mass of Alum. Because the molar mass of any molecule (or compound) can be calculated by simply adding the molar masses of individual atoms. I hope you have understood the short and simple calculation for finding the molar mass of Na2SO4. If you have a periodic table with you, then you can easily calculate the molar mass of Alum KAl(SO4)2 Hence the Molar mass of Na2SO4 is 142.036 g/mol. So, Molar mass of Na2SO4 = Molar mass of 2 Sodium (Na) atoms + Molar mass of 1 Sulfur (S) atom + Molar mass of 4 Oxygen (O) atoms. Prepare a concept map and use the proper conversion factor. You can see that in Na2SO4, there are 2 Sodium atoms, 1 Sulfur atom and 4 Oxygen atoms. 
Final Exam General Studies Science: Help & Review Status: Not Started. We created a timeline of the history of the periodic table.Now, to calculate the molar mass of Na2SO4, you just have to add the molar mass of all the individual atoms that are present in Na2SO4. Understand how the molar mass is important to chemical experiments Calculate grams using molar mass Practice Exams. The Periodic Table has constantly been improved and developed over the past 200 years, but in 1869 Dimitri Mendeleev finished the first version of the periodic table as we know it today, by arranging the elements by atomic mass and leaving spaces open for the elements that were not yet discovered. Who invented the Periodic Table of Elements? You can see that The molar mass of Sodium is 22.990 g/mol. So let’s look at the molar mass of Sodium, Sulfur and Oxygen from the above periodic table. Now in Na2SO4, there are 2 Sodium atoms, 1 Sulfur atom and 4 Oxygen atoms. The periodic table also gives us an idea of what the characteristics of an element might be and help us predict how an element might react based on in which group it is located. Immediately find any imaginable information about an element, calculate molar masses of any compounds you like, facilitate your overview with infographics and much more. You can see the molar mass value of all the atoms from this periodic table. /PeriodicTableoftheElements-5c3648e546e0fb0001ba3a0a.jpg)
Remember thats the number written under the element symbol and element name. So in most cases, to find the molar mass of an element, you just need to look at its atomic mass (atomic weight) on the periodic table. The Periodic Table of Elements can be used as an assisting tool in chemical calculations, when a specification of an element is needed it is easily found in the Periodic Table. The molar mass of an element is the mass in grams of one mole (6.02 x 10 23 particles) of the element. How is the Periodic Table of Elements used? The table lists all the elements that are currently known (118), in descending order of the number of protons that are present, in a single atom of the element. The Periodic table of elements is a tool, developed by scientists over hundreds of years. NaCl has one of each atom 22.990 + 35.453 58.443 g in one mole of NaCl H 2 0. Sodium (Na) Molar Mass 22.99 g Magnesium (Mg) g. A mole is a unit of measurement that is used to express the amount of a substance in chemistry. List of all the elements and their properties: On the periodic table, the atomic mass given is the mass (in grams) of one mole of that atom.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
